
Multiple Equilibria- If a reaction can be expressed as the sum of two or more reactions, the equilibrium constant for the overall reaction is given by the product of the equilibrium constants of the individual reactions Reaction Quotient (Qc)- For reactions that have not reached equilibrium, we obtain the reaction quotient (Qc), instead of the equilibrium constant by substituting the initial concentrations into the equilibrium constant expression. Homogeneous Equilibrium- applies to reactions in which all reacting species are in the same phase. Heterogeneous Equilibrium- results from a reversible reaction involving reactants and products that are in different phases. Law of Mass Action- holds that for a reversible reaction at equilibrium and a constant temperature, a certain ratio of reactant and product concentrations has a constant value, K (the equilibrium constant). Physical Equilibrium- changes that occur are physical processes. Relationship between ∆G and ∆G° Chemical Equilibrium- is achieved when the rates of the forward and reverse reactions are equal and the concentrations of the reactants and products remain constant. ∆G is the change of Gibbs (free) energy for a system and ∆G° is the Gibbs energy change for a system under standard conditions. At that point the RT ln Q term will become negative enough to match the positive ∆G° term in magnitude. Thus, the net reaction will proceed from right to left until a significant amount of reactant has been formed. Case 2: A large positive ∆G° term will tend to make ∆G also positive. At that point, the RT ln Q term will become positive enough to match the negative ∆G° term in magnitude. Thus, the net reaction will proceed from left to right until a significant amount of product has been formed. Case 1: A large negative value of ∆G° will tend to make ∆G also negative. 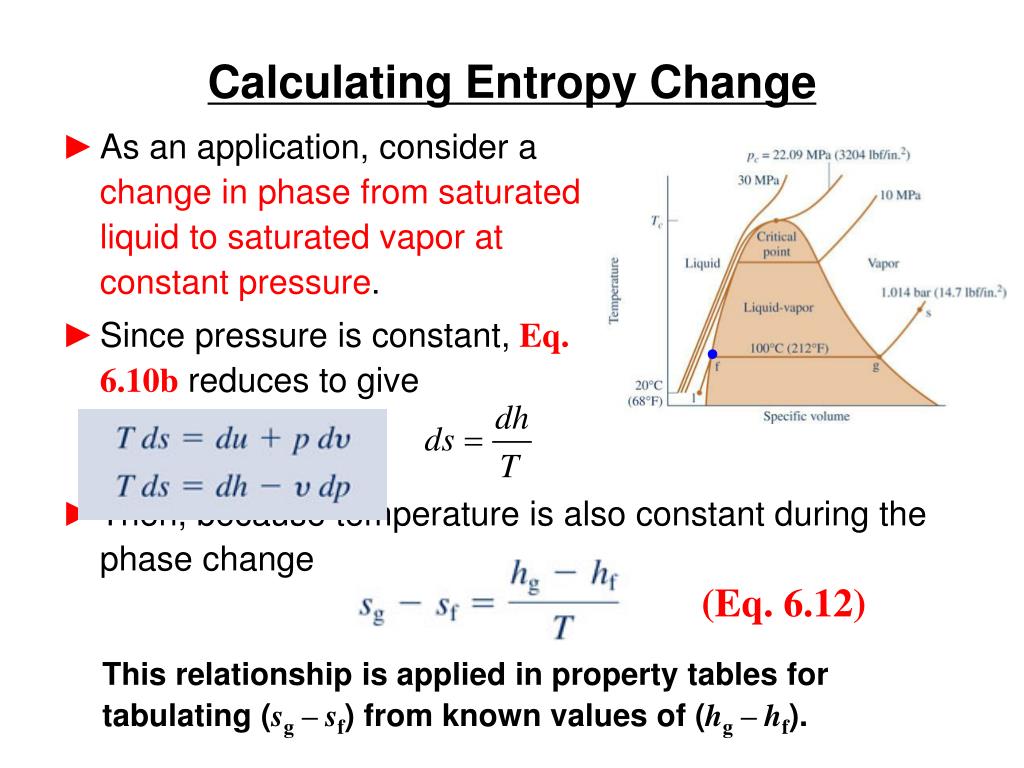 
Gibbs Free Energy/ Free Energy- express the spontaneity of a reaction more directly. If there is no net change in the total number of gas molecules, then ∆S° may be positive or negative, but will be relatively small numerically. If the total number of gas molecules diminishes, ∆S° is negative. Standard Entropy of a reaction- given by the difference in standard entropies between products and reactants: If a reaction produces more gas molecules than it consumes, ∆S° is positive. Standard Entropy- is the absolute entropy of a substance at 1 atm and 25☌. If ∆H is negative and ∆S is negative, then ∆G will be negative only when T∆S is smaller in magnitude than ∆H. If ∆H is negative and ∆S is positive, then ∆G will always be negative regardless of temperature. A system with more microstates (larger W) among which to spread its energy (large dispersal) has a higher entropy. Change in Entropy: o A system with fewer microstates (smaller W) among which to spread its energy (small dispersal) has a lower entropy. 
Entropy- a measure of how spread out or dispersed the energy of a system is among the different possible ways that system can contain energy. Spontaneous Reaction- a reaction that occurs when reactants are brought together under a specific set of conditions (for example, at a certain temperature, pressure, and concentration). Third Law of Thermodynamics- the entropy of a perfect crystalline substance is zero at the absolute zero of temperature. Second Law of Thermodynamics- The entropy of the universe increases in a spontaneous process and remains unchanged in an equilibrium process. First Law of Thermodynamics- states that energy can be converted from one form to another, but cannot be created nor destroyed. State of a function- properties that are determined by the state of the system, regardless of how that condition was achieved. State of a System- is defined by the values of all relevant macroscopic properties, for example, composition, energy, temperature, pressure, and volume. Thermodynamics- the scientific study of the interconversion of heat and other kinds of energy.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
